Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

McGrady, J.; Kumagai, Yuta; Watanabe, Masayuki; Kirishima, Akira*; Akiyama, Daisuke*; Kimuro, Shingo; Ishidera, Takamitsu
Journal of Nuclear Science and Technology, 60(12), p.1586 - 1594, 2023/12
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology) and CrUO
and CrUO
Akiyama, Daisuke*; Kusaka, Ryoji; Kumagai, Yuta; Nakada, Masami; Watanabe, Masayuki; Okamoto, Yoshihiro; Nagai, Takayuki; Sato, Nobuaki*; Kirishima, Akira*
Journal of Nuclear Materials, 568, p.153847_1 - 153847_10, 2022/09
Times Cited Count:3 Percentile:68.71(Materials Science, Multidisciplinary)FeUO , CrUO
, CrUO , and Fe
, and Fe Cr
Cr UO
UO are monouranates containing pentavalent U. Even though these compounds have similar crystal structures, their formation conditions and thermal stability are significantly different. To determine the factors causing the difference in thermal stability between FeUO
are monouranates containing pentavalent U. Even though these compounds have similar crystal structures, their formation conditions and thermal stability are significantly different. To determine the factors causing the difference in thermal stability between FeUO and CrUO
and CrUO , their crystal structures were evaluated in detail. A Raman band was observed at 700 cm
, their crystal structures were evaluated in detail. A Raman band was observed at 700 cm in all the samples. This Raman band was derived from the stretching vibration of the O-U-O axis band, indicating that Fe
in all the samples. This Raman band was derived from the stretching vibration of the O-U-O axis band, indicating that Fe Cr
Cr UO
UO was composed of a uranyl-like structure in its lattice regardless of its "x"' value. M
was composed of a uranyl-like structure in its lattice regardless of its "x"' value. M ssbauer measurements indicated that the Fe in FeUO
ssbauer measurements indicated that the Fe in FeUO and Fe
and Fe Cr
Cr UO
UO were trivalent. Furthermore, Fe
were trivalent. Furthermore, Fe Cr
Cr UO
UO lost its symmetry around Fe
lost its symmetry around Fe with increasing electron densities around Fe
with increasing electron densities around Fe , as the abundance of Cr increased. These results suggested no significant structural differences between FeUO
, as the abundance of Cr increased. These results suggested no significant structural differences between FeUO and CrUO
and CrUO . Thermogravimetric measurements for UO
. Thermogravimetric measurements for UO , FeUO
, FeUO , and CrUO
, and CrUO showed that the temperature at which FeUO
showed that the temperature at which FeUO decomposed under an oxidizing condition (approximately 800
decomposed under an oxidizing condition (approximately 800  C) was significantly lower than the temperature at which the decomposition of CrUO
C) was significantly lower than the temperature at which the decomposition of CrUO started (approximately 1250
started (approximately 1250  C). Based on these results, we concluded that the decomposition of FeUO
C). Based on these results, we concluded that the decomposition of FeUO was triggered by an "in-crystal" redox reaction, i.e., Fe
was triggered by an "in-crystal" redox reaction, i.e., Fe
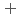 U
U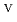
 Fe
Fe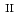
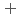 U
U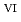 , which would not occur in the CrUO
, which would not occur in the CrUO lattice because Cr
lattice because Cr could never be reduced under the investigated condition. Finally, the existence of Cr
could never be reduced under the investigated condition. Finally, the existence of Cr in FexCr
in FexCr UO
UO effectively suppressed the decomposition of the Fe
effectively suppressed the decomposition of the Fe Cr
Cr UO
UO crystal, even at a very low Cr content.
crystal, even at a very low Cr content.
Cantarel, V.; Yamagishi, Isao
Journal of Nuclear Science and Technology, 59(7), p.888 - 897, 2022/07
Times Cited Count:2 Percentile:53.91(Nuclear Science & Technology)Arai, Yosuke*; Kuroda, Kenta*; Nomoto, Takuya*; Tin, Z. H.*; Sakuragi, Shunsuke*; Bareille, C.*; Akebi, Shuntaro*; Kurokawa, Kifu*; Kinoshita, Yuto*; Zhang, W.-L.*; et al.
Nature Materials, 21(4), p.410 - 415, 2022/04
Times Cited Count:7 Percentile:77.62(Chemistry, Physical)Saeki, Morihisa*; Yomogida, Takumi; Matsumura, Daiju; Saito, Takumi*; Nakanishi, Ryuzo*; Tsuji, Takuya; Oba, Hironori*
Analytical Sciences, 36(11), p.1371 - 1378, 2020/11
Times Cited Count:2 Percentile:10(Chemistry, Analytical)We measured X-ray absorption fine structure (XAFS) and Raman spectra of isopolymolybdates(VI) in HNO solution (0.15- 4.0 M), which change their geometries depending on acid concentration, and performed simultaneous resolution of the XAFS and Raman data using a multivariate curve resolution by alternating least-squares (MCR-ALS) analysis. In iterative ALS optimization, initial data matrices were prepared by two different methods. The MCR-ALS result of single XAFS data matrix shows large dependence on the preparation method of the initial data matrices. The MCR-ALS result of an augmented matrix of Raman and XAFS data has little dependence on the initial data matrices. It indicates that the augmentation method effectively improves the rotation ambiguities in the MCR-ALS analysis of the XAFS data. Based on the model fitting of the pure EXAFS oscillations, we revealed the change of [Mo
solution (0.15- 4.0 M), which change their geometries depending on acid concentration, and performed simultaneous resolution of the XAFS and Raman data using a multivariate curve resolution by alternating least-squares (MCR-ALS) analysis. In iterative ALS optimization, initial data matrices were prepared by two different methods. The MCR-ALS result of single XAFS data matrix shows large dependence on the preparation method of the initial data matrices. The MCR-ALS result of an augmented matrix of Raman and XAFS data has little dependence on the initial data matrices. It indicates that the augmentation method effectively improves the rotation ambiguities in the MCR-ALS analysis of the XAFS data. Based on the model fitting of the pure EXAFS oscillations, we revealed the change of [Mo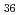 O
O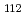 (H
(H O)
O)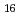 ]
]
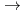 [Mo
[Mo O
O (H
(H O)
O) ]
]
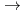 [HMoO
[HMoO (H
(H O)
O) ]
] in the highly concentrated HNO
in the highly concentrated HNO solution.
solution.
 MoO
MoO , in severe accident conditions, 1; Partitioning of Cs and Mo among gaseous species
, in severe accident conditions, 1; Partitioning of Cs and Mo among gaseous speciesDo, Thi Mai Dung*; Sujatanond, S.*; Ogawa, Toru
Journal of Nuclear Science and Technology, 55(3), p.348 - 355, 2018/03
Times Cited Count:6 Percentile:52.79(Nuclear Science & Technology)In order to better understand the behavior of cesium in the severe accident of the LWR, the high-temperature chemistry of Cs MoO
MoO in H
in H O+H
O+H gas was studied. The pseudo-binary system, Cs
gas was studied. The pseudo-binary system, Cs MoO
MoO -MoO
-MoO , was thermochemically modeled with Redlich-Kister formulation to form a basis to analyze the high-temperature behavior of Cs
, was thermochemically modeled with Redlich-Kister formulation to form a basis to analyze the high-temperature behavior of Cs MoO
MoO . The model prediction was compared with the thermogravimetric measurements of Cs
. The model prediction was compared with the thermogravimetric measurements of Cs MoO
MoO in dry and humid argon, which revealed that the mass-loss rate was enhanced in the humid atmosphere. Thermochemical model was further applied to predict the partitioning of cesium and molybdenum among gaseous species in the BWR core degradation condition typical of Short-Term Station Blackout.
in dry and humid argon, which revealed that the mass-loss rate was enhanced in the humid atmosphere. Thermochemical model was further applied to predict the partitioning of cesium and molybdenum among gaseous species in the BWR core degradation condition typical of Short-Term Station Blackout.
Nagai, Takayuki; Sato, Nobuaki*; Sasage, Kenichi
Busshitsu, Debaisu Ryoiki Kyodo Kenkyu Kyoten Kenkyu Seika Hokokusho (Heisei-27-Nendo) (CD-ROM), 2 Pages, 2016/03
no abstracts in English
Ikeda, Takashi
Journal of Chemical Physics, 141(4), p.044501_1 - 044501_8, 2014/07
Times Cited Count:15 Percentile:51.43(Chemistry, Physical)From both the polarized and depolarized Raman scattering spectra of supercritical water a peak located at around 1600 cm , attributed normally to bending mode of water molecules, was experimentally observed to vanish, whereas the corresponding peak remains clearly visible in the measured infrared (IR) absorption spectrum. In this computational study a theoretical formulation for analyzing the IR and Raman spectra is developed via first principles molecular dynamics combined with the modern polarization theory. We demonstrate that the experimentally observed peculiar behavior of the IR and Raman spectra for water are well reproduced in our computational scheme. We discuss the origins of a feature observed at 1600 cm
, attributed normally to bending mode of water molecules, was experimentally observed to vanish, whereas the corresponding peak remains clearly visible in the measured infrared (IR) absorption spectrum. In this computational study a theoretical formulation for analyzing the IR and Raman spectra is developed via first principles molecular dynamics combined with the modern polarization theory. We demonstrate that the experimentally observed peculiar behavior of the IR and Raman spectra for water are well reproduced in our computational scheme. We discuss the origins of a feature observed at 1600 cm in Raman spectra of ambient water.
in Raman spectra of ambient water.
 thin films by ion irradiation
thin films by ion irradiationNarumi, Kazumasa; Sakai, Seiji; Naramoto, Hiroshi*; Takanashi, Koki
JAEA-Review 2005-001, TIARA Annual Report 2004, p.238 - 240, 2006/01
no abstracts in English
 films polymerized by ion irradiation
films polymerized by ion irradiationNarumi, Kazumasa; Sakai, Seiji; Naramoto, Hiroshi*; Takanashi, Koki
Fullerenes, Nanotubes, and Carbon Nanostructures, 14(2-3), p.429 - 434, 2006/00
no abstracts in English
Gordon, E. B.*; Kumada, Takayuki; Ishiguro, Masazumi; Aratono, Yasuyuki
Journal of Experimental and Theoretical Physics, 99(4), p.776 - 783, 2004/10
The solid Deuterium clusters for the first time isolated in a matrix of solid Helium have been investigated at T = 1.3K and P = 3 MPa by CARS (Coherent anti-Stokes Raman spectroscopy) technique. The Q1(J=0) and Q1(J=1) lines intensity, shape and positions have been studied as the functions of ortho- para content in the solid, as well as of clusters size. The strong effect of Raman scattering cross section sensitivity to the molecular environment nuclear spin state has been found in CARS; the ratio of probabilities for the scattering by para (J=1) and ortho (J=0) deuterium being equal to 1 in a gas is as high as 10,000 in nearly pure o-D2, whereas it is about 50 in spontaneous Raman scattering. This effect has been shown to give rise starting from cluster size corresponding to the phonon band onset.
Kohara, Shinji*; Suzuya, Kentaro; Takeuchi, Ken*; Loong, C.-K.*; Grimsditch, M.*; Weber, J. K. R.*; Tangeman, J. A.*; Key, T. S.*
Science, 303(5664), p.1649 - 1652, 2004/03
Times Cited Count:158 Percentile:96.05(Multidisciplinary Sciences)Inorganic glasses normally exhibit a network of interconncted covalent-bonded structural elements that has no long-range order. In silicate glasses the network formers are based on SiO -tetrahedra interconnected via oxygen atoms at the corners. Conventional wisdom then implies that alkaline and alkaline-earth orthosilicate materials cannot be vitrified because they do not contain sufficient network forming SiO
-tetrahedra interconnected via oxygen atoms at the corners. Conventional wisdom then implies that alkaline and alkaline-earth orthosilicate materials cannot be vitrified because they do not contain sufficient network forming SiO to establish the needed interconnectivity. We have studied a bulk magnesium orthosilicate glass obtained by containerless melting-and-cooling. We find that the role of network former is largely taken on by corner- and edge-sharing of highly distorted ionic Mg-O species that adopt 4-, 5- and 6-coordination with oxygen. The results suggest that similar novel glassy phases may be found in the containerless environment of interstellar space.
to establish the needed interconnectivity. We have studied a bulk magnesium orthosilicate glass obtained by containerless melting-and-cooling. We find that the role of network former is largely taken on by corner- and edge-sharing of highly distorted ionic Mg-O species that adopt 4-, 5- and 6-coordination with oxygen. The results suggest that similar novel glassy phases may be found in the containerless environment of interstellar space.
Sakai, Seiji; Naramoto, Hiroshi; Xu, Y.; Priyanto, T. H.; Lavrentiev, V.; Narumi, Kazumasa
Materials Research Society Symposium Proceedings, Vol.788, p.L11.49.1 - L11.49.6, 2004/00
Mixture films between cobalt and C (CoxC
(CoxC , x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C
, x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C -based phase in which certain number of cobalt atoms are coordinated with C
-based phase in which certain number of cobalt atoms are coordinated with C molecules. It is deduced that the equilibrated number of cobalt atoms in the C
molecules. It is deduced that the equilibrated number of cobalt atoms in the C -based phase is 4 atoms per C
-based phase is 4 atoms per C molecule and in which an electron transfer occurs from a cobalt atom to a C
molecule and in which an electron transfer occurs from a cobalt atom to a C molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x
molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x  4 are thermally activated-type, and their conductive nature can be attributed to the C
4 are thermally activated-type, and their conductive nature can be attributed to the C -based phase. Further (2) the mixtures between 4
-based phase. Further (2) the mixtures between 4  x
x  60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x
60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x  60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
Assefa, Z.*; Yaita, Tsuyoshi; Haire, R. G.*; Tachimori, Shoichi
Inorganic Chemistry, 42(23), p.7375 - 7377, 2003/11
Times Cited Count:11 Percentile:36.73(Chemistry, Inorganic & Nuclear)The 6-methyl- 2-(2-pyridyl)-benzimidazole (biz) ligand coordinates with the actinide species in solution, and the complexes display efficient intra-molecular energy-transfer processes. The energy transfer in the Cm(III):biz system proceeds in a non-radiative mode, whereas a radiative mode is the principal mechanism in the Am(III)-biz system.
 -irradiated diamond
-irradiated diamondXu, Y.; Naramoto, Hiroshi; Narumi, Kazumasa; Miyashita, Kiyoshi*; Kamiya, Tomihiro; Sakai, Takuro
Applied Physics Letters, 83(10), p.1968 - 1970, 2003/09
Times Cited Count:6 Percentile:28.18(Physics, Applied)no abstracts in English
Soga, Takeshi
Spectrochimica Acta, Part A, 59(11), p.2497 - 2510, 2003/09
no abstracts in English
Kato, Chiaki
JAERI-Research 2003-013, 143 Pages, 2003/08
This study is investigation about stress corrosion cracking (SCC) of zirconium in nuclear fuel reprocessing. Chapter 1 is described background. Chapter 2 is explained experimental apparates. Chapter 3 is described the increased oxidization potential on the heat-transfer surface and suggested the initiation of SCC on a boiling heat-transfer surface. Chapter 4 is described that the SCC susceptibility increased with increasing nitric acid concentration and solution temperature on notched specimen by SSRT. In addition, the SCC susceptibility effected by the crystal anisotropy by the hot rolling direction and increased on a parallel face to the rolling direction. Chapter 5 is described that the SCC susceptibility increased in HAZ/base metal boundary in order to the preferential orientation of cleavage plane (0002). Chapter 6 is described that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles.
Wei, P.; Xu, Y.; Nagata, Shinji*; Narumi, Kazumasa; Naramoto, Hiroshi
Nuclear Instruments and Methods in Physics Research B, 206(1-4), p.233 - 236, 2003/05
Times Cited Count:6 Percentile:43.07(Instruments & Instrumentation)no abstracts in English
Lavrentiev, V.; Abe, Hiroaki; Yamamoto, Shunya; Naramoto, Hiroshi; Narumi, Kazumasa
Surface and Interface Analysis, 35(1), p.36 - 39, 2003/01
Times Cited Count:8 Percentile:22.7(Chemistry, Physical)no abstracts in English
Naramoto, Hiroshi; Xu, Y.; Narumi, Kazumasa; Zhu, X.; Vacik, J.; Yamamoto, Shunya; Miyashita, Kiyoshi*
JAERI-Review 2001-039, TIARA Annual Report 2000, p.183 - 185, 2001/11
no abstracts in English